Galvanic corrosion is the result of an electrochemical reaction. For galvanic corrosion to take place, four things must exist simultaneously: an anode, a cathode, an electrolyte and a conductive path between two pieces of metal. A galvanic circuit is created in which the anode loses electrons to the cathode with the assistance of the electrolyte. The galvanic cell created by two dissimilar metals and the presence of the electrolyte operates only in one direction. Consequently, the anode eventually disintegrates.
In PV installations, the anode and cathode consist of metals, such as stainless steel, copper and aluminum. Water commonly serves as the electrolyte. Whether galvanic corrosion is a serious problem depends on the potential failure point. For a PV installation, the long-term effects of corrosion can range from unsightly finishes to racking or fastener failure.
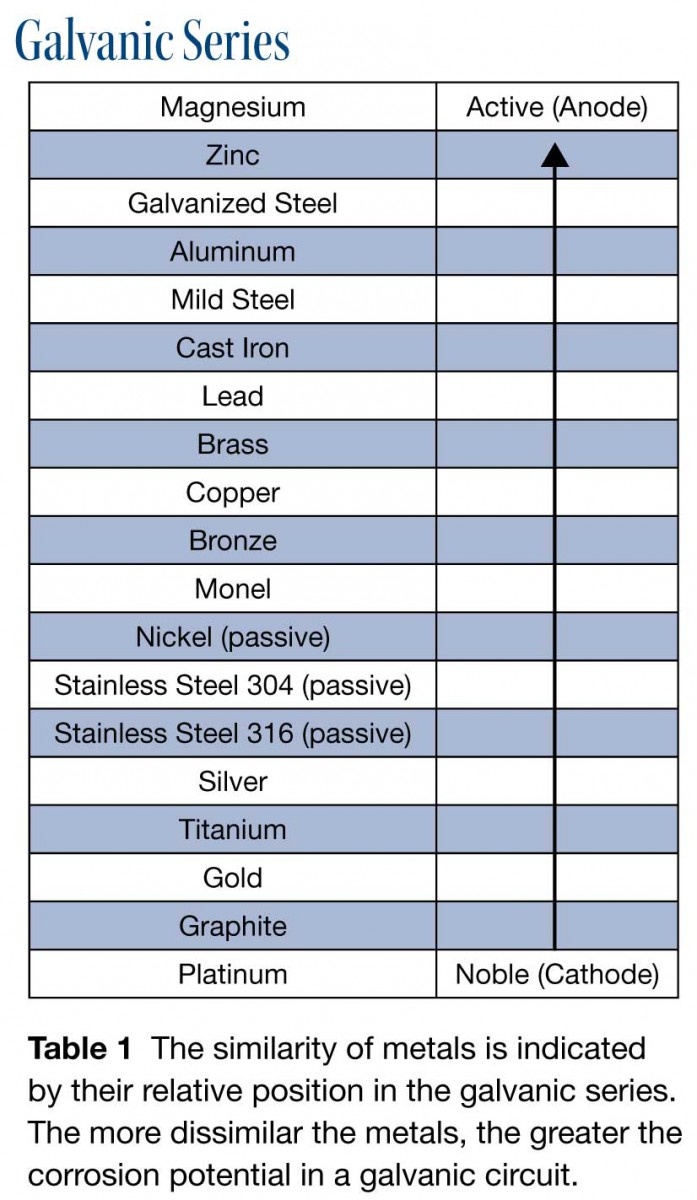
The more dissimilar the metals, as reflected by their relative position in the galvanic series, the greater the corrosion potential in the galvanic circuit. The general rule is to avoid joining metals far apart in the galvanic series. For example, steel is anodic next to brass, and stainless steel is cathodic next to zinc or aluminum. Another way to read this is that steel corrodes next to brass and stainless steel, while aluminum and zinc corrode next to steel when an electrolyte and a conductive path are present.
Every metal has a standard electrical potential (voltage) based on its ability to release or accept electrons when in contact with a dissimilar metal and an electrolyte. In reality, the galvanic system is more dynamic than most published material on voltage-potential data suggests. The actual reaction that takes place between two metals in the environment is dependent on electrolyte concentration, pH, temperature and other factors. Rob Haddock from Metal Roof Innovations, manufacturer of the S-5! mounting clamp, provides a word of caution about using the galvanic scale. «Some installers might want to use the galvanic scale to identify dissimilar metals, but the graphical galvanic scale is not always a good way to determine whether one metal is compatible with another,» he says. «When metals oxidize, the oxide layer created is a new material that may or may not exhibit the electrochemical characteristics of the parent material.»
More detailed information is currently available in Ukrainian language.